South America
The content of this page was informed by Danilo Fernandez Rios and Florencia Goberna.
This page provides a single portal for key gene editing regulatory documents across South America.
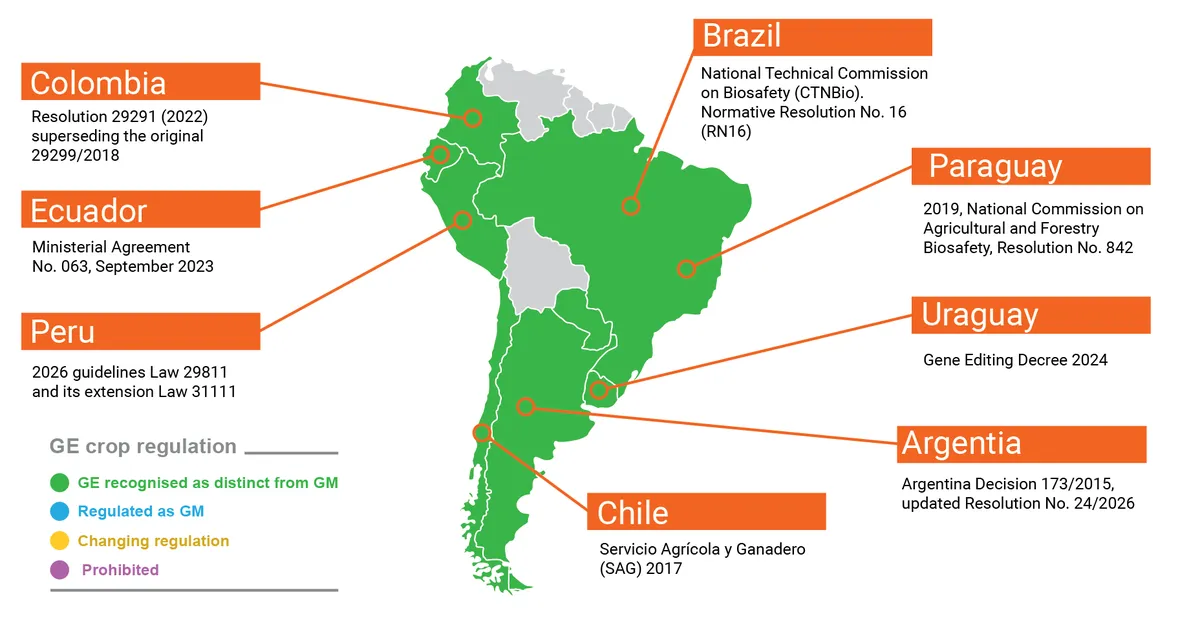
Argentina:
Key documents: Ministry of Economy, Secretariat of Agriculture, Livestock and Fisheries. Resolution No. 24/2026.
A full description of the policy and regulatory developments across Argentina can be found in this post by Florencia Goberna.
Argentina was the first country in the world to establish a regulatory framework specifically for gene-edited crops, through Decision 173/2015 of the Ministry of Agriculture, Livestock and Fisheries (MAGyP). The framework requires developers to submit a dossier to the Argentine Biosafety Commission (CONABIA), which determines on a case-by-case basis whether the product contains stable foreign DNA. Gene-edited products without foreign DNA are classified as conventional and exempt from GMO regulation. This product-based, foreign-DNA criterion has since become the near-universal international regulatory standard, and Argentina's approach is widely referred to in the literature as the "Argentina model."
Brazil:
Key document: Ministry of Science, Technology, Innovation, and Communications. Resolution Nº 16/2018. Alternative link.
Brazil's National Technical Commission on Biosafety (CTNBio) established its framework for gene-edited crops through Normative Resolution No. 16 (RN16), issued 15 January 2018 and published in the Diário Oficial da União on 22 January 2018. RN16 defines a category of "Innovative Precision Improvement Techniques" (TIMPs) — encompassing gene editing and related NBTs — and establishes a consultation procedure through which developers can determine whether their product should be classified as a genetically modified organism (OGM) or as a conventional product. Products without recombinant DNA/RNA in the final product are eligible for non-GMO classification. In December 2024, MARA issued five biosafety certificates for gene-edited events, including the first gene-edited rice event approved for domestic cultivation, marking a significant operational milestone for the framework.
Chile
Key document: Ministry of Agriculture (2026: still open for public consultation).
Chile's gene editing regulatory framework operates under Resolution No. 1523/2001 of the Servicio Agrícola y Ganadero (SAG), which governs living modified organisms. Since 2017, SAG has operated an informal case-by-case evaluation procedure under this resolution to determine whether NBT-derived plant material constitutes a living modified organism or a conventional product, based on whether it contains a new combination of genetic material. A draft resolution published for public consultation on 7 January 2026 — Resolución Exenta: Establece Requisitos Técnicos y Administrativos para Determinar Normativa Aplicable al Material Vegetal de Propagación Desarrollado por Nuevas Técnicas de Mejoramiento (NBT) — would formalise this procedure as a mandatory administrative process, with a defined 20 working-day determination timeline. Notably, the draft explicitly excludes insertions of alleles from the same species from the definition of "new combination of genetic material." As of May 2026 this draft has not yet entered into force, pending publication in the Diario Oficial.
Colombia
Key document: Ministry of Agriculture and Rural Development, Colombian Institute of Agriculture and Livestock. Resolution No. 00022991/2022. Link here, but not available from UK.
Colombia's gene editing regulatory framework is administered by the Instituto Colombiano Agropecuario (ICA), operating under the Ministry of Agriculture and Rural Development. The original framework was established by ICA Resolution No. 29299/2018, which created a case-by-case procedure for determining whether cultivars developed through precision biotechnology techniques should be classified as Living Modified Organisms (OVMs) or as conventional organisms. This was superseded and extended by ICA Resolution No. 00022991 (11 November 2022), which broadened the scope of the procedure to cover animals of zootechnical interest in addition to crop plants. The central regulatory criterion is product-based: only products containing stable foreign genetic material are classified as OVMs. Products without foreign DNA are treated as conventional and regulated accordingly under standard seed and livestock frameworks.
Ecuador
Key document: Ministry of Agriculture, Livestock, and Fisheries. Resolution N° 063/2023.
Ecuador's approach to gene editing regulation operates within a constitutionally constrained framework. Article 401 of the 2008 Constitution declares Ecuador a GMO-free country, creating a structural ceiling on regulatory reform. Within this constraint, the Organic Code of the Environment (2019) and subsequent ministerial instruments established that only organisms containing recombinant or foreign DNA qualify as GMOs under Ecuadorian law — effectively creating a definitional exemption for transgene-free gene-edited products without overriding the constitutional prohibition. This was operationalised through Ministerial Agreement No. 063, signed by the Minister of Agriculture and Livestock on 13 September 2023, which approved a Technical Guide for the use of seeds and crops obtained by precision breeding techniques. The first application under this framework — for a gene-edited banana variety developed in response to Fusarium wilt TR4 pressure — was submitted in October 2024. The constitutional constraint means Ecuador's exemption rests on definitional exclusion rather than explicit legislative authorisation, creating ongoing structural fragility.
Paraguay:
Key document: Ministry of Agriculture and Livestock. Resolution N° 842/2019.
Paraguay established its gene editing regulatory framework in 2019 through Resolution No. 842 of the National Commission on Agricultural and Forestry Biosafety (CONBIO). The resolution established requirements for the evaluation of crops and other products developed using gene editing and other new breeding techniques, with classification determined on a case-by-case basis according to whether the final product contains foreign DNA. Paraguay was among the earlier Latin American adopters of a GE-specific regulatory pathway, following Argentina, Brazil, and Chile.
Peru
Key document: Ministry of Environment. Decree No. 000068/2026.
Peru's gene editing regulatory situation is distinctive in the region due to the coexistence of a new GE classification framework with an active GMO moratorium. Law 29811 (2011), extended by Law 31111 to 31 December 2035, maintains a moratorium on the entry and production of Living Modified Organisms (LMOs). Within this moratorium framework, Peru's Ministry of the Environment (MINAM) approved Ministerial Resolution No. D000068-2026-MINAM-DM on 8 March 2026, establishing technical and scientific criteria to determine whether products or organisms developed through new breeding techniques (including gene editing) fall within the scope of the moratorium as LMOs. Transgene-free gene-edited products that do not meet the LMO definition may be classified as conventional, but this legal clarity does not confer cultivation rights — commercial deployment of gene-edited crops remains constrained by the moratorium until at minimum 2035. Peru therefore has regulatory clarity without operational freedom, a combination that distinguishes it from other green-coded countries in the region.
Uruguay
Key document: Ministry of Livestock, Agriculture, and Fisheries Ministry of Environment. Decree 84/024.
Uruguay established its gene editing regulatory framework through a Gene Editing Decree announced on 18 March 2024, making it one of the more recent Latin American adopters of a GE-specific regulatory pathway. The decree established a Technical Working Group process to determine on a case-by-case basis whether a gene-edited product should be classified as a GMO, with the central criterion being the presence or absence of foreign DNA in the final product. Uruguay's framework is consistent with the broader regional convergence around the product-based foreign-DNA criterion that has emerged across Latin America since Argentina's pioneering 2015 decision.