Ghana Ghana allows GE for cultivation, import and export Conventional-like
Ghana's National Biosafety Authority (NBA) have published guidelines for Genome Editing Applications under Section 40(3) of the Biosafety Act, 2011 (Act 831). The guidance exempts gene edited organims from Ghana's existing GMO legislation and provides secondary instruments for the existing biosafety law.
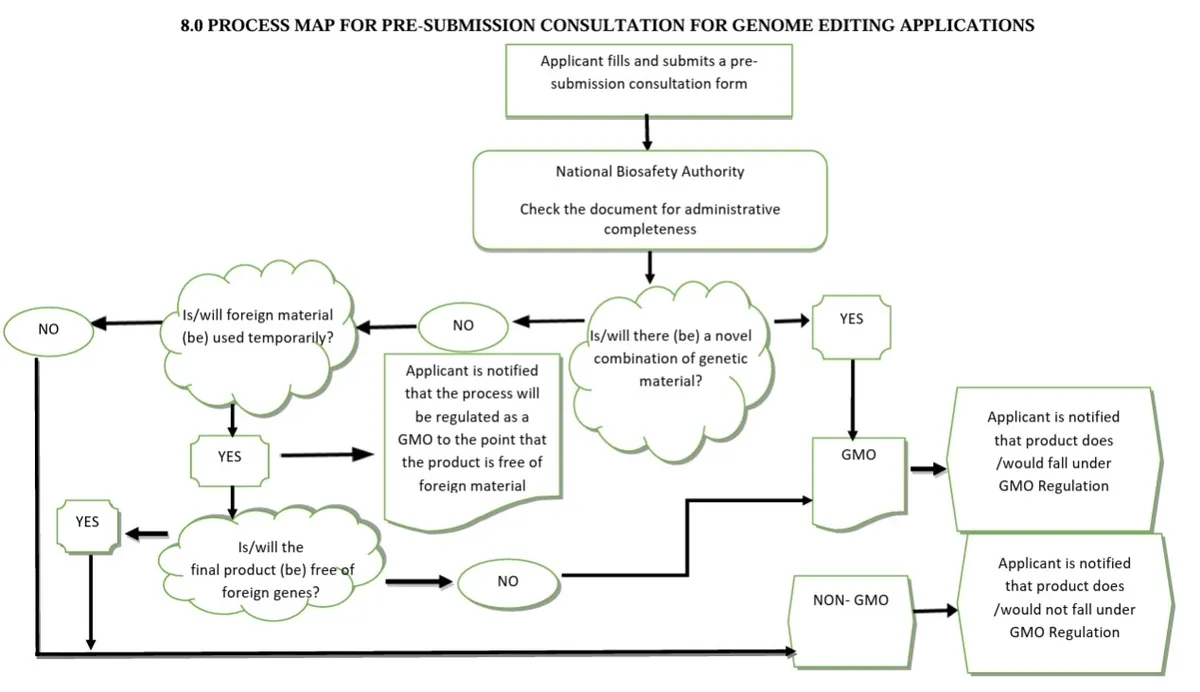
The guidelines rule that GE products containing genes from non-sexually compatible species will be reguated under the GMO act, but that transgene-free products will be exempt from this act. Instead they will be regulated on a case-by-case basis as conventionally-bred products.
End products from SDN-1 methods that do not contain inserted nucleic acids are exempt from the GMO act regulation. SDN-2-type changes are not specifically mentioned, but would likely be regulated as convention if the final product does not contain genes from outside those that are sexually compatible with the species.
Applicants complete a pre-submission consultation form to the secretariat, which helps determine the regulatory status of the application. If the product is regulated under the Act, the applicant then submits a formal application to the CEO of the NBA, and the NBA may request further information or clarification. A flow chart is provided to guide this determination.